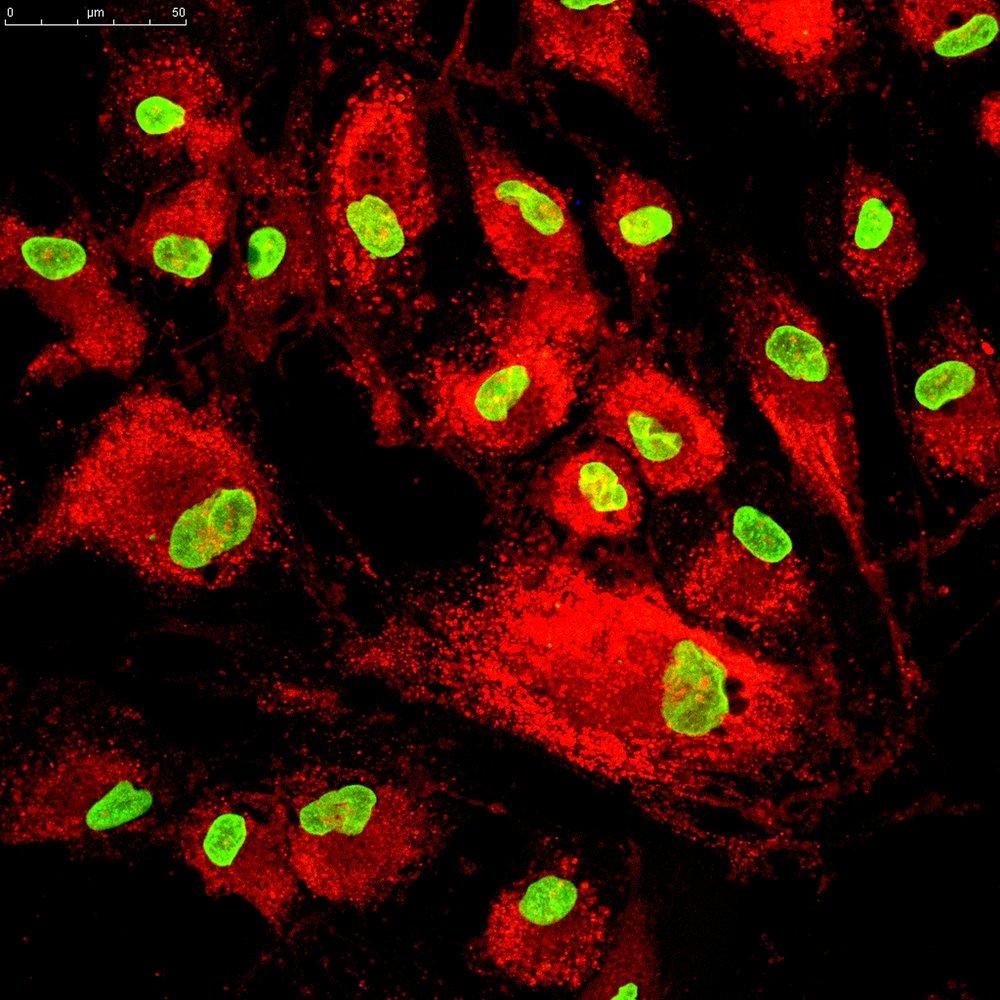
Transchymal-UC are mesenchymal stem cells (MSCs), multipotent stromal cells that can differentiate in vitro into a variety of cell types including osteoblasts (bone), chondrocytes (cartilage), adipocytes (fat). Transchymal-UC can self-renew and have a high proliferative capacity. Transchymal-UC cells are harvested from freshly umbilical cord tissue, and have functional and phenotypic similarities with bone-marrow derived MSCs. As with all mesenchymal stem cells, these cells are characterised by the expression of cell-surface markers SSEA4, CD90, CD105, and CD73, and by the absence of CD34, CD14, CD19, CD45; they are plastic adherent when maintained in standard culture conditions.
Transchymal-UC cells are expanded in culture and configured as per the format required. Cells are produced in batches, and each batch, or lot, originates from a single bio-discard (or umbilical cord); they are ready-to-use, and no sub culturing culturing procedure is required.
Care is taken to ensure the safety of the donor and the integrity of collected biosample, and the handling procedures are compliant with Institutional Ethics Committee (IEC). Donors are tested for infection by HIV-1, HIV-2, HBV, HCV, Mycoplasma, Bacteria, Yeast and Fungi before collection of the biosample. Cells are processed batch wise to be bioburden free.
Configured Transchymal-UC cell systems have application in exploratory preclinical evaluations such as high throughput screening (HTS) of test materials, drugs, chemicals, or biologics, for efficacy, cytotoxicity, and haematotoxicity. Transchymal-UC is also suitable for developing humanised 3D bioorgans (bone, cartilage, and wound) and models.
Disclaimer: Product for in vitro research use only. Not approved for diagnostic, therapeutic, or clinical applications. Not approved for human or veterinary use in vivo.









Reviews
There are no reviews yet.